|
Eg for MgCl2 it would be equal to Mg + 2 x Cl 24.305 + (2 x 35.45) 95. For a molecule or compound, simply add up all the molar masses of the elements, taking subscripts into account. The relative atomic mass is the same as the molar mass (except molar mass is in g/mol). The molar mass of any element is on the periodic table. Do this by looking up sodium (Na) on the periodic table. For example, find the mass of one mole of sodium. energy E molar energy Em wavenumber J eV Eh kJ. Example 1: Find the Molar Mass of an Element. Add them together: add the results from step 3 to get the total molar mass of the compound. IUPAC 2005 standard atomic weights (mean relative atomic masses) as approved at the 43rd IUPAC General Assembly in Beijing, China in August 2005 are listed with uncertainties in the last figure in parentheses M. 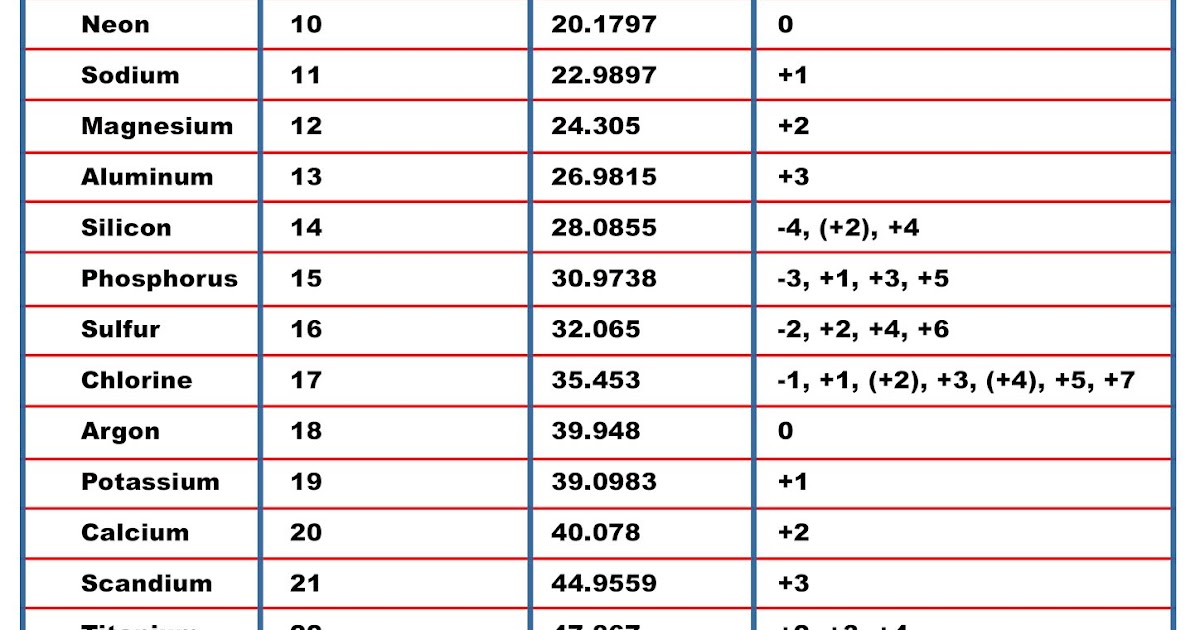
Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu).  
For normal samples from earth with typical isotope composition, the atomic weight can be approximated by the standard atomic weight or the conventional atomic weight. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, M u 1 × 10 3 kgmol 1. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
