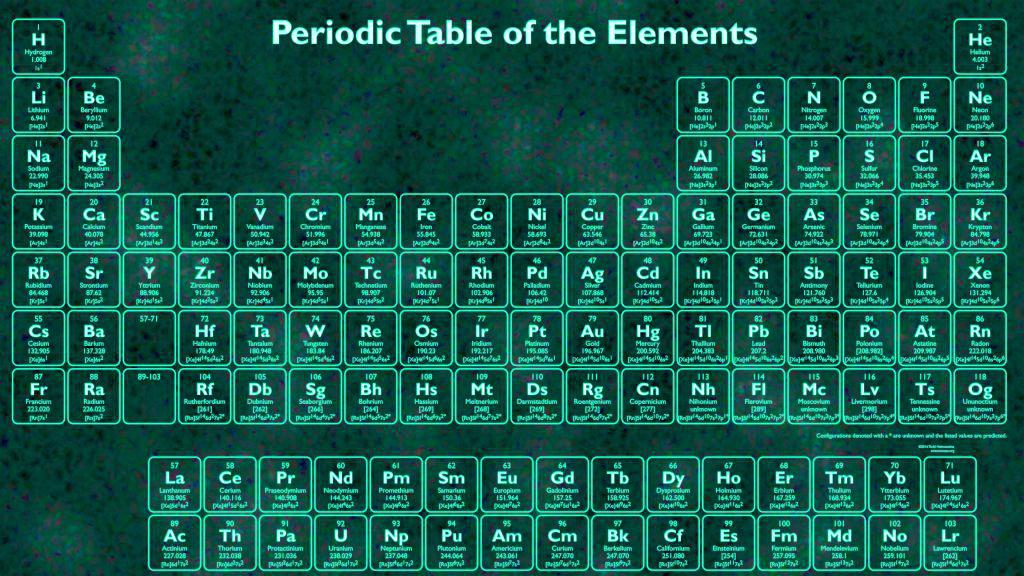
The first periodic table to become generally accepted was that of the Russian chemist Dmitri Mendeleev in 1869 he formulated the periodic law as a dependence of chemical properties on atomic mass. Nonmetallic character increases going from the bottom left of the periodic table to the top right. Metallic character increases going down a group and decreases from left to right across a period. Vertical, horizontal and diagonal trends characterize the periodic table. Elements in the same group tend to show similar chemical characteristics. The table is divided into four roughly rectangular areas called blocks. It is a depiction of the periodic law, which states that when the elements are arranged in order of their atomic numbers an approximate recurrence of their properties is evident. It is an icon of chemistry and is widely used in physics and other sciences. A 10 minute time limit is imposed on the advanced students.The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows (" periods") and columns (" groups"). These students are directed to not use the periodic table, and are challenged to search for the elements based only on their prior knowledge. Advanced Level: these instructions are intended for advanced students who are very familiar with element names, specifically the first 20 elements as well as other common elements.These students are given a 15 minute time limit to find as many element names as possible. Students are told to use the periodic table as a resource for finding element names, as well as told to focus on the first 20 elements. Intermediate Level: these instructions are intended for intermediate students (middle school or regular level chemistry high school) with some familiarity of the periodic table.Students are given a word bank, and are not held to a time restriction, although a teacher may choose to impose one. Beginner Level: these instructions are intended for younger students (elementary or middle school) with little chemistry background knowledge.This activity includes three puzzles, each with differentiated instructions so that it can be used at a variety of grade levels.An answer key document has been provided for teacher reference. Some names overlap throughout the puzzle, sharing common letters. Names are written both horizontally and vertically, as well as forward and backward. There are 30 element names hidden in the word search.Alternatively it might be used during or at the end of a unit studying the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
